
Pirudhan Karak, Ph.D.
Department of Chemistry
Indian Institute of Science Education and Research, Bhopal, India
Research Areas
(I) Metal Catalyzed C–H Functionalization
Transition metal-catalyzed C-H activation catalysis stands at the forefront of modern organic synthesis. This powerful tool allows for direct functionalization of carbon-hydrogen bonds, streamlining synthetic pathways and reducing step counts. We explored the C-H activation/functionalization at the Metal-NHC (N-heterocyclic Carbene) platform. We developed a novel double rollover π-extension (RoPE) strategy to access π-conjugated organic molecules via multiple C-H activation in a one-pot reaction. The synergy between catalysis, mechanistic understanding, and method development represents an exciting frontier in synthetic organic chemistry, promising further advancements and transformative applications in the creation of complex organic molecules.
Selected papers:
Chem. Commun. 2019, 55, 6791-6794,
Chem. Commun. 2022, 58, 133-154
Angew. Chem. Int. Ed. 2023, e202310603


Describe your image


(II) Sustanable Catalysis: Bio-inspired Electrochemical CO2 Reduction and HER
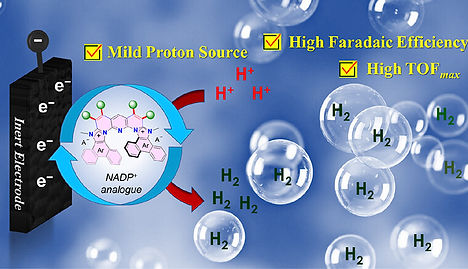
Organic cofactors such as nicotinamide adenine dinucleotide phosphate (NADH) and reduced flavin adenine dinucleotide (FADH2) involve various enzymatic redox reactions in the natural photosynthesis process. In chemistry, the synthetic analogs of these cofactors have been successfully utilized in organic synthetic chemistry, such as regio-and enantioselective reduction of organic substrates (i.e., imines, carbonyl compounds) to their corresponding reduced products in a stoichiometric manner. However, catalytic use of these synthetic NADH analogs is still challenging due to the regeneration of active reduced species in the reaction conditions. Toward this direction, we introduced a novel class of helicene-based organic hydride donors. Then, as a proof-of-principle, these organic hydride donors were successfully employed for the catalytic electrochemical CO2 reduction reaction (eCO2RR) process which showed a significantly improved activity with more than stoichiometric turnover featuring a 100−1000-fold enhancement of the existing TON values. Later, the same system was employed for the metal-free electrochemical hydrogen evolution reaction (HER) using weak acid as a proton source at a moderate overpotential and the system showed a high Faradaic efficiency and TOF values of 80% for H2 improvement over the existing organic system
Selected papers:
J. Am. Chem. Soc. 2023, 145, 13, 7230–7241
J. Am. Chem. Soc. 2023, 145, 31, 17321–17328

(III) Supramolecular chemistry: Functional organic molecules, heterohelicenes
π-Conjugated molecules represent a cornerstone in the realm of optoelectronic applications due to their unique electronic properties. These molecules find diverse applications in organic solar cells, light-emitting diodes (LEDs), organic field-effect transistors (OFETs), and sensors. The tunability of their optical and electronic properties through molecular engineering allows for precise control over energy levels, bandgaps, and exciton dynamics, laying the groundwork for highly efficient and versatile optoelectronic devices. In this context, we applied double rollover π-extension (RoPE) protocol to access stimuli-responsive (pH, light) heteroaromatic compounds. We have also synthesized a series of [9]-azahelicenes, enabled with stimuli (acid/base and light)-responsive modules. We have demonstrated for the first time the reversible spring-like behaviour of the flexible heterohelicenes experimentally as well as through computational analysis.
Selected papers:
Chem. Sci., 2022, 13, 11163-11173
Chem. Commun. 2019, 55, 6791-6794

